|
10/25/2023 0 Comments Complete ionic equation  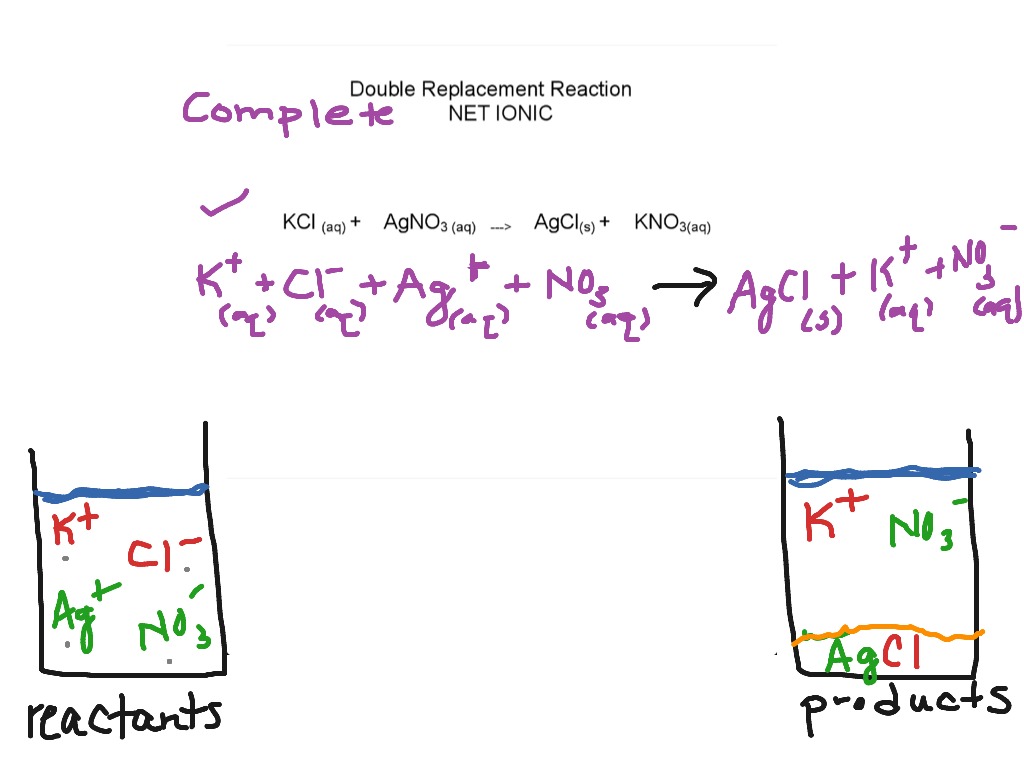
Reaction between sodium hydroxide and sulphuric acid to form sodium sulphate and water. (Spectator ions are ions that do not take part in the reaction) Spectator ions are omitted from the ionic equation.The key to being able to write net ionic. +3 on left must have +3 on right as well) Net ionic equations are useful in that they show only those chemical species participating in a chemical reaction. As a diligent student of chemistry, you will likely encounter tons of reactions that occur in aqueous solution (perhaps you are already drowning in them). Total charges carried by the ions must be balanced (e.g.Write the balanced net ionic equation to describe any reaction that occurs when the solutions of Na 2 SO 4 and NH. Write three equations (complete chemical equation, complete ionic equation, and net ionic equation) that describe this process. Number of atoms of each elements must be balanced When a solution of AgNO 3 is added to a solution of CaCl 2, insoluble AgCl precipitates.Recall that ionic compounds that dissolved in water will dissociate completely into ions (have charge). This video covers, how to predict products, how to balance a chemical equation, how to identify the solubility of a compound, how to write a complete ionic e. It is just there to balance out the charge because you can't have an ion just by itself - you must always pair an ion with another one of opposite charge so that the overall charge is zero.Writing ionic equation is extremely similar to writing chemical equations. Write the ionic equation by breaking all the soluble ionic compounds (those marked with an (aq)) into their respective ions. Notice that the Cl- was canceled out from the Net Ionic Equation, because it really isn't playing an important part of this reaction. Enter an equation of an ionic chemical equation and press the Balance button. Net ionic equation: a molecular equation that depicts the ions or molecules that experience a change within a reaction. Step 3: Finally, the ionic formula for the ionic compound will be displayed in the new window. Complete ionic equation: a molecular equation that separates the molecules into their ion forms. Step 2: Now click the button Ionic Formula to get the result. This is called the Total Ionic Equation.ģ) Then, you simplify by canceling things out if they appear on both sides of the reaction, resulting in the Net Ionic Equation.Īnd other oxidation half and simply add themįor example, let look at the reaction of calcium carbonate with hydrochloric acid to form calcium chloride, water, and carbon dioxide:ĬaCO3(s) + 2HCl(aq) -> CaCl2(aq) + H2O(l) + CO2(g)ĬaCO3(s) + 2H+(aq) + 2Cl-(aq) -> Ca2+(aq) + 2Cl-(aq) + H2O(l) + CO2(g)ĬaCO3(s) + 2H+(aq) -> Ca2+(aq) + H2O(l) + CO2(g) How to Use the Ionic Calculator The procedure to use the ionic calculator is as follows: Step 1: Enter the ionic compound name in the input field. 
Step 2: Complete and net ionic equations (b). Write the reaction out with all of the separated ions. The net ionic equation is a short representation of only those ions involved in the physical or chemical changes. If something is insoluble, it should not be broken apart. You will have to use the solubility rules to do this, they can be found online. HCl(aq) + NaOH(aq) -> NaCl(aq) + H2O() Only full formulas (never words or ions) are involved in a complete molecular equation. This is also called the Molecular Equation.Ģ) Then, you break apart the soluble molecules into the two ions that are formed (one positive and one negative). Find more Chemistry widgets in WolframAlpha. Complete ionic equations show all the atoms in a chemical equation whereas net ionic equations do not show the ions that cancel out from both sides of the eqn. To write a Net Ionic Reaction, follow these 3 steps:ġ) Start by simply writing the overall balanced chemical reaction. Get the free 'NET IONIC EQUATION CALCULATOR' widget for your website, blog, Wordpress, Blogger, or iGoogle. just the names of the reactants Total Ionic Equation: Ca2+(aq)+ 2 Cl- (aq) + 2 Na+ (aq)+ CO32- (aq) -> 2 Na+(aq) + 2 Cl-(aq) + CaCO Net Ionic Equation: Ca2+(. A Net Ionic Equation is a chemical equation for a reaction which lists only those species participating in the reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
